However, this ideal geometry is not usually seen because there is a lot of space underneath the pyramid. It would be straight above the blue atom and at 90 ° to all of the other atoms. In addition, there would be a fifth atom forming the top point or the apex of the pyramid. In an ideal square pyramid, there would be a square planar base with one central atom (blue) and four atoms arranged around it at 90 °. The bond angles in these two geometries are slightly more complicated than in the earlier cases. A fifth atom occupies the space above the In thisĬase, the central atom sits above a base of four neighbors those four positionsįorm a square plane together. The other common five coordinate geometry is square pyramidal. Planar positions referred to as "equatorial" the other two positions are called Two neighbours are roughly colinear with the central atom. The last two neighbours are above and below this plane. In a trigonal bipyramidal structure, theĬentral atom is surrounded by three neighbours that form a trigonal plane around In the case of five neighboring atoms, there are two geometries thatĪre roughly equally prevalent. So, a tetrahedral geometry is described as having 109 ° bond angles but a square planar geometry is described as having 90 ° bond angles. That puts them all a little closer to each other. In a squre planar geometry, those four atoms all share the same plane. In a tetrahedron, the four red atoms are arranged in three dimensions around the central blue atom. Note that the bond angles in a square planar geometry are not the same as in a tetrahedron. This geometry is most often observed forĬertain compounds of palladium and platinum as well as a few other transition Planar geometry, all four neighbours are found in the same plane and are roughlyĩ0 degrees apart from each other. Second geometry is available for a limited number of compounds. Is the most common geometry for tjis coordination number. That could beĪdvantageous because it minimizes crowding. Seen, maximizes the distances between neighbours. There are actually two different geometric options that occur with some We can think about the geometry around a particular Remember, the coordination number is just the number of atoms in close contact 
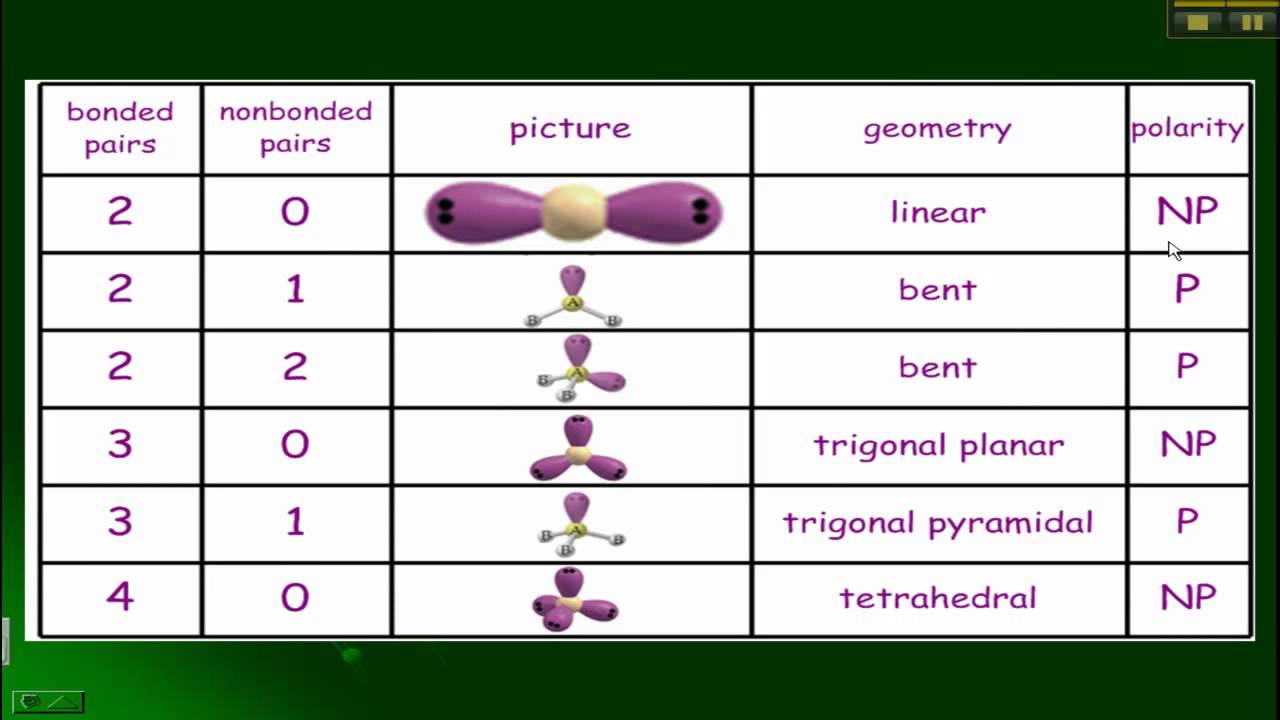
Structures and considered the "coordination number" of the atom or ion. Some other molecular geometries that are pretty common.Įarlier, we looked at the environments of atoms or ions within solid These geometries are found throughout the periodic table for atoms that haveĢ, 3 or 4 neighbouring atoms within a molecule. It makes sense that as more atoms are attached to the central atom, they crowd a little more closely together, so the angle between them gets smaller. In the tetrahedral structure, the angle between two of the bonds between the blue atom and any two red atoms is always 109.5 °. By comparison, in the linear structure, two red atoms are found 180 ° apart, so the atoms are spaced a little farther apart in the linear structure than in the trigonal planar structure. We call this the bond angle it's the angle formed by going from one of the red atoms to the central blue atom to a second red atom. The angle at each corner would be 60 °, but from the internal perspective of the molecule, it's more useful to know how widely spaced two red atoms are when they are attached to the same blue atom. Assuming they are arranged at equal distances around the blue atom, they would form an equilateral triangle. For example, in the trigonal planar geometry shown here, if viewed from above, the three red atoms would form the corners of a triangle. 
The same aspects of geometry show up in molecules, although we are often concerned with the angles formed at the intersection of two bonds where they meet an atom, rather than the corners of the shapes described by the atoms. The term "geometry" often brings to mind angles in shapes such as triangles and squares. Or three neighbouring atoms, and so linear or trigonal planar geometries are Carbon most often has four neighbouring atoms in the molecule,Īnd in that case it is tetrahedral. 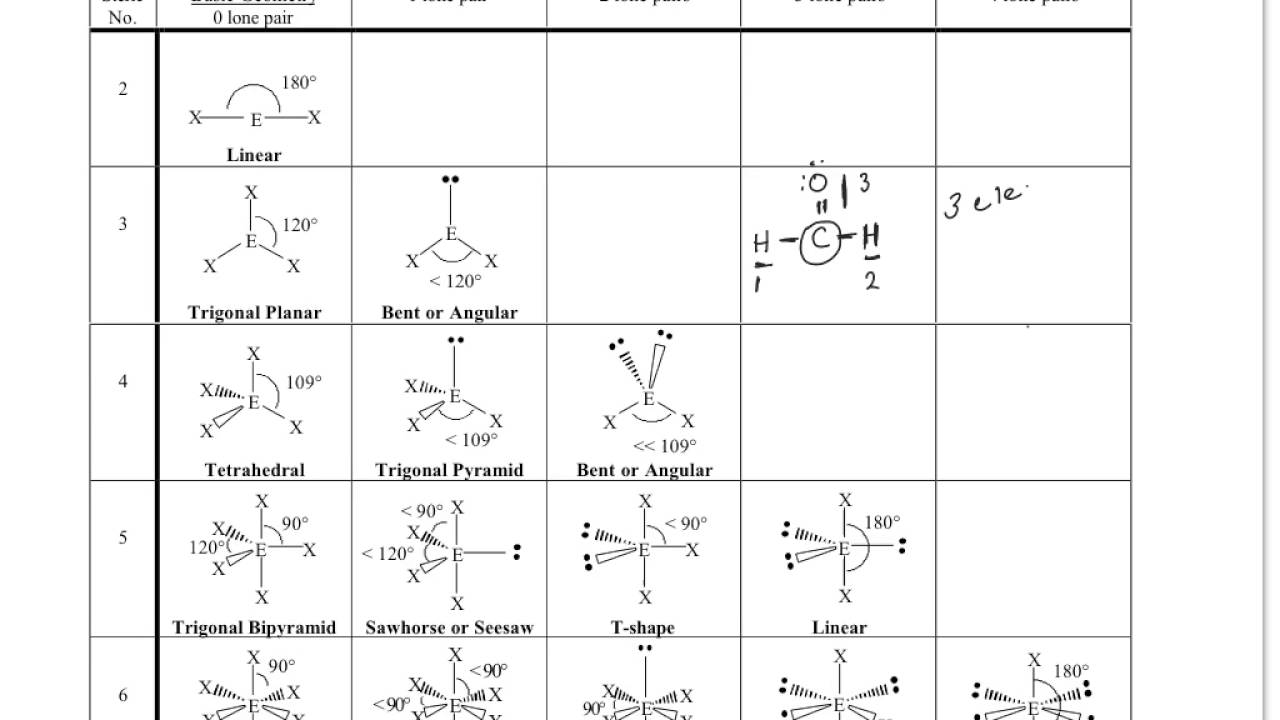
Previously, we looked at a few common geometries of carbon atoms in
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
